 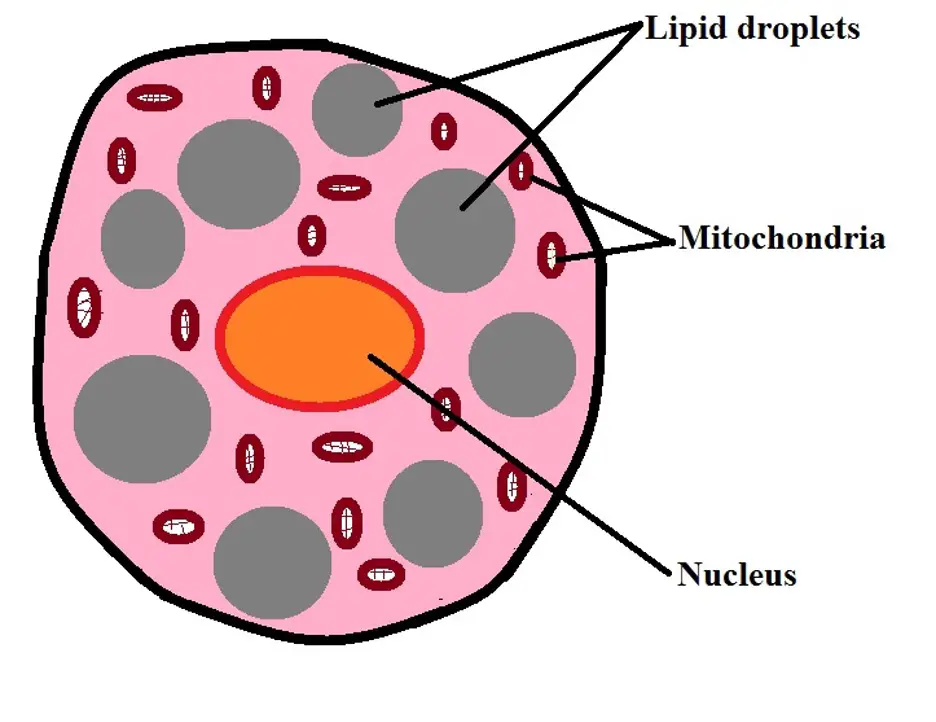 
WAT, however, also contributes to metabolic dysregulation that characterizes insulin resistance and obesity-related metabolic and cardiovascular complications 2, 3.īrown adipose tissue (BAT), enriched in mitochondria, regulates adaptive thermogenesis in small rodents and mammalian newborns. In addition, via production of endocrine factors, adipocytokines, and lipids, WAT regulates and integrates important physiological pathways, including satiety, energy utilization, glucose sensitivity, insulin sensitivity, and inflammation 1. White adipose tissue (WAT), a highly regulated and dynamic secretory organ, affects body fat and energy utilization through storage and turnover/hydrolysis of triglycerides. Obesity is a major risk factor for type 2 diabetes and cardiovascular disease. Results reported here suggest that PDE3B and/or its downstream signaling partners might be important regulators of energy metabolism in adipose tissue, and potential therapeutic targets for treating obesity, diabetes and their associated metabolic disorders. In epididymal white adipose tissue (eWAT) of PDE3B KO mice on a SvJ129 background, cAMP/protein kinase A (PKA) and AMP-activated protein kinase (AMPK) signaling pathways are activated, resulting in “browning” phenotype, with a smaller increases in body weight under high-fat diet, smaller fat deposits, increased β-oxidation of fatty acids (FAO) and oxygen consumption. To study effects of cyclic nucleotides on energy homeostatic mechanisms, mice were generated by targeted inactivation of cyclic nucleotide phosphodiesterase 3b (Pde3b) gene, which encodes PDE3B, an enzyme that catalyzes hydrolysis of cAMP and cGMP and is highly expressed in tissues that regulate energy homeostasis, including adipose tissue, liver, and pancreas. Cyclic adenosine monophosphate (cAMP) is very important in the development of the beige phenotype and activation of its thermogenic program. 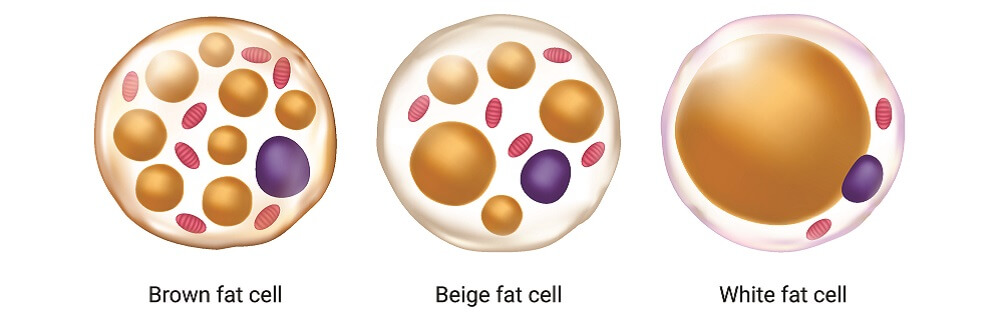
The authors have declared no competing interest.Understanding mechanisms by which a population of beige adipocytes is increased in white adipose tissue (WAT) reflects a potential strategy in the fight against obesity and diabetes. In summary, this study indicates that aging blocks beige adipogenesis and dysregulates adipocyte responses to cold exposure and provides a unique resource for identifying cold and/or aging-regulated pathways in adipose tissue. We further identified natriuretic peptide clearance receptor Npr3, a beige fat repressor, as a marker gene for a subset of white adipocytes and an aging-upregulated gene in adipocytes. Notably, cold exposure induced an adipocyte population expressing high levels of de novo lipogenesis (DNL) genes, and this response was severely blunted in aged animals. Examination of adipocytes by single nucleus RNA-sequencing identified compositional and transcriptional differences in adipocyte populations with age and cold exposure. Fibroblastic ASPC populations from young and aged mice were equally competent for beige differentiation in vitro, suggesting that environmental factors suppress adipogenesis in vivo. We found that aging increases the expression of Cd9 and other fibrogenic genes in fibroblastic ASPCs and blocks their differentiation into beige adipocytes. Here, we evaluate the impact of aging on the profile and activity of adipocyte stem and progenitor cells (ASPCs) and adipocytes during the beiging process. The energy-burning capability of beige adipose tissue is a potential therapeutic tool for reducing obesity and metabolic disease, but this capacity is decreased by aging. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
